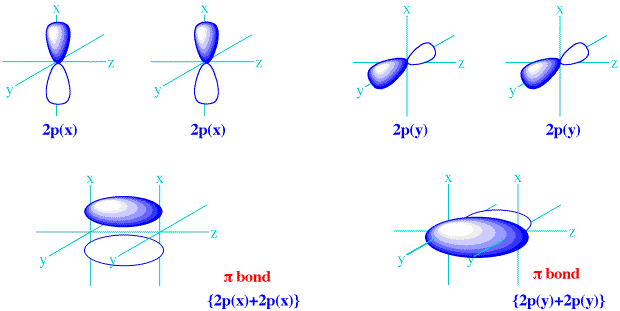
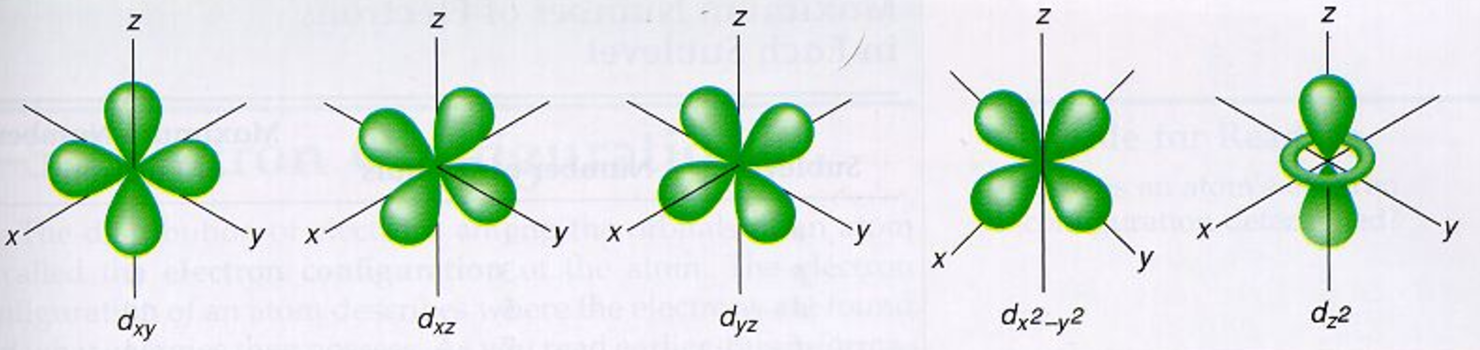
DEGENERATE ATOMIC ORBITALS FULL
Here we demonstrate that realistic magnesium, zinc, and cadmium clusters having a specific tetrahedral framework possess anomalous higher-fold degeneracies than spherical symmetry from first principles. In addition, by means of simple tight-binding models and group-theoretical analyses, we elucidate that these degeneracies can be attributed to dynamical symmetry.ĭensity functional theory (DFT) calculations were performed for T d symmetrical structures with valence electron numbers sufficient for the full occupation of superatomic orbitals. However, it is expected that species with this type of symmetry could exceed the spherical symmetry of atoms and exhibit unique electronic and magnetic properties. With the exception of the extreme example of the hydrogen atom, such dynamical symmetry has not yet been found in an actual substance. This occurs because the Hamiltonian of this oscillator has U(3) symmetry 1, 3, 12, 14. Another example is the three-dimensional isotropic harmonic oscillator, in which the 2 S and 1 D orbitals are 6-fold degenerate, the 2 P and 1 F orbitals are 10-fold degenerate, and the 3 S, 2 D, and 1 G orbitals are 15-fold degenerate, and so on. As a result, the atom formally possesses four-dimensional spherical symmetry. A typical example is the hydrogen atom, in which the unoccupied atomic orbitals are highly degenerate, because its Hamiltonian possesses symmetry associated with the Laplace–Runge–Lenz vector 13, in addition to three-dimensional spherical symmetry. As this symmetry originates not from geometrical properties but rather from dynamical characteristics, it is referred to as dynamical symmetry. Non-geometrical symmetry has also been known, which gives rise to a greater degree of orbital degeneracy than that of spherical symmetry O(3) 3, 12. The degree of degeneracy must be less than six based on point-group theory 11, with the exception of spin-orbital degeneracy.

However, even if a cluster belongs to the highest point group, I h, its superatomic orbitals will split depending on their irreducible representations. The spherical jellium model has been applied with considerable success in cluster science.
Various other examples have also been reported, including Au 20 having T d symmetry 10. The molecular orbitals of this cluster are superatomic S, P, D, F, ⋯ orbitals and a stable closed-shell structure is formed similar to that of a halogen anion. The most outstanding example is an aluminum cluster, Al 13 −, having I h symmetry 4. Clusters satisfying the magic number requirement have been detected and characterized by gas-phase spectroscopic methods. As the full occupation of superatomic orbitals results in stable closed shells, the numbers of valence electrons necessary for closed shells are termed magic numbers. This analogy can be understood based on the three-dimensional spherical jellium model 9. A superatom is analogous to an atom but with a higher-order structure: highly symmetrical metal clusters possess delocalized molecular orbitals, the shapes of which are just like those of atomic orbitals. Recently, the concept of a superatom has been proposed and developed 4, 5, 6, 7, 8. It is of interest to consider whether or not it is possible to overcome this limitation. However, species having higher degrees of degeneracy than atoms have not yet been known, due to the limitations of geometrical symmetry. This degeneracy in turn leads to certain properties, such as closed-shell stability and magnetism. As a result of this symmetry, these species possess atomic orbitals with the high degrees of degeneracy, such as d orbitals, which have a fivefold degeneracy. In real space, atoms have the highest geometrical symmetry, three-dimensional spherical symmetry O(3). Symmetry is the most fundamental concept in both physics and chemistry 1, 2, 3, and the basic properties of classical and quantum systems can be derived based on this concept.
